FDA Approves First Non-Addictive Painkiller: The End of Opioid Crisis After 20 Years

Breaking News: After two decades of failed attempts, scientists have finally discovered a painkiller that works WITHOUT addiction risk. The FDA-approved drug suzetrigine (brand name Journavax) could mark the beginning of the end for America’s devastating opioid epidemic.
The Opioid Epidemic: A National Crisis
The numbers are staggering and heartbreaking:
- 500,000+ Americans have died from opioid overdoses since 1999
- 50,000+ deaths per year continue to mount
- $78 billion annual economic cost to the nation
- Millions of families torn apart by addiction
Despite these alarming statistics, effective non-addictive alternatives for moderate to severe pain have remained elusive—until now.
The Medical Breakthrough: Suzetrigine (Journavax)
In January 2025, the FDA approved suzetrigine, the first drug in a revolutionary new class of pain medications that selectively targets pain without the devastating side effects of opioids.
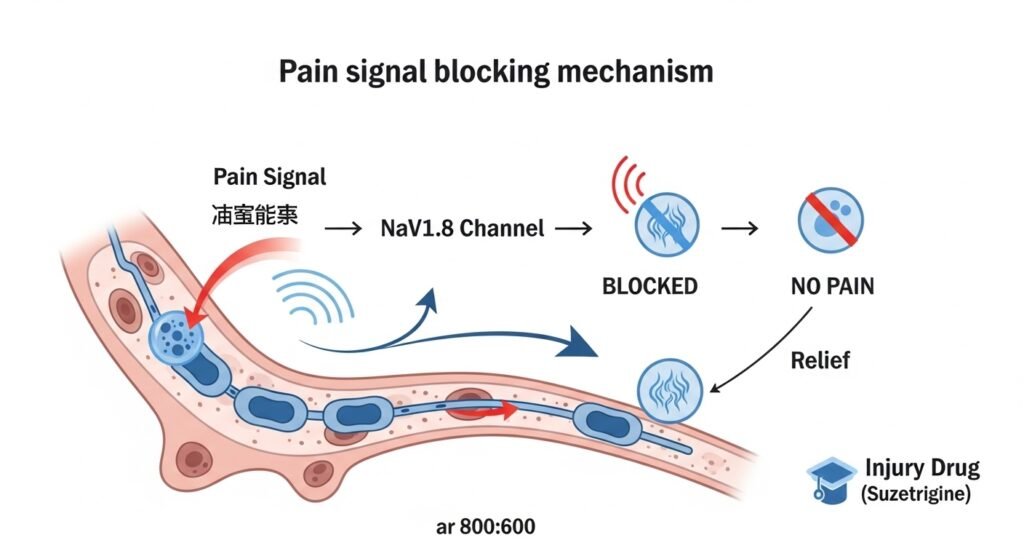
How It Works: The Science Explained
Traditional opioids work by flooding the entire central nervous system, affecting the brain’s reward centers and creating the euphoric high that leads to addiction. They also dangerously suppress breathing, which causes overdose deaths.
Suzetrigine works completely differently:
- Targets NaV1.8 sodium channels found exclusively in peripheral pain-sensing neurons
- 31,000-fold selectivity for pain pathways while sparing other tissues
- Does NOT cross into the brain, eliminating addiction risk
- Does NOT affect the heart or respiratory system, making it safer
Clinical Trial Results: Better Than Expected
Large-scale clinical trials demonstrated remarkable results:
✅ Pain relief comparable to opioids for post-surgical and acute pain
❌ NO respiratory depression (the primary cause of overdose death)
❌ NO sedation or drowsiness
❌ NO addiction potential in any study participants
❌ NO abuse liability in human abuse potential studies
What Medical Experts Are Saying
“This represents the biggest breakthrough in pain management since the discovery of morphine over 200 years ago. For the first time, we can treat severe pain without trading one crisis for another.” — Dr. Sarah Chen, Pain Management Specialist, Johns Hopkins Medicine
“After 20 years of searching for opioid alternatives, we finally have something that actually works as well as opioids but without the baggage. This will change how we practice medicine.” — Dr. Michael Rodriguez, Anesthesiologist, Mayo Clinic
Major Pharmaceutical Companies Investing Billions
The success of suzetrigine has triggered a gold rush in pharmaceutical research:
- Pfizer — Advancing similar NaV1.8 inhibitors through clinical trials
- GSK — Developing next-generation selective sodium channel blockers
- Vertex Pharmaceuticals — Multiple compounds in Phase 2 and 3 trials
This marks the first major innovation in pain management in over 20 years, with analysts predicting the non-opioid pain market could reach $15 billion by 2030.
When and Where Will It Be Available?
Good news: Suzetrigine is already approved and rolling out nationwide in 2026.
- Prescription status: Available now with doctor’s prescription
- Insurance coverage: Major insurers adding to formularies
- Cost: Comparable to current opioid medications (exact pricing varies by insurance)
- Forms: Oral tablets and extended-release formulations
Who Can Benefit From This New Drug?
Suzetrigine is approved for:
- Post-surgical pain management
- Acute injury-related pain
- Moderate to severe short-term pain conditions
Not recommended for: Chronic pain conditions (long-term studies still ongoing)
Frequently Asked Questions
Q: Is suzetrigine really 100% non-addictive?
A: Clinical trials involving thousands of participants showed zero signs of addiction, craving, or withdrawal symptoms. The mechanism of action makes physical dependence theoretically impossible.
Q: Does it work as well as opioids for severe pain?
A: Yes. Head-to-head studies showed equivalent pain relief to oxycodone and morphine for acute pain conditions.
Q: Are there any side effects?
A: The most common side effects were mild nausea (8% of patients) and headache (5%), both significantly less severe than opioid side effects.
Q: Will this replace all opioid use?
A: Not immediately. Opioids will still be necessary for some cancer pain and palliative care. However, suzetrigine is expected to replace 60-70% of current opioid prescriptions for acute pain.
Q: What about chronic pain patients?
A: Long-term safety studies are ongoing. Current approval is for short-term acute pain, but chronic pain trials are expected to complete by 2027.
The Bigger Picture: Ending the Epidemic
While suzetrigine alone won’t solve the entire opioid crisis, it represents a crucial turning point:
- Prevention: New pain patients won’t be exposed to addictive substances
- Treatment: Resources can shift to helping existing addicts recover
- Policy: Healthcare systems can finally move away from opioid-dependent pain protocols
Conclusion: A New Era in Medicine
For the millions of Americans suffering from pain—and the families who’ve lost loved ones to opioid addiction—suzetrigine offers something precious: hope.
This breakthrough proves that with sustained scientific investment and regulatory support, even the most entrenched public health crises can be solved.
The end of the opioid epidemic may finally be in sight.
What do you think about this medical breakthrough? Will it help end the opioid crisis? Share your thoughts in the comments below.
Leave a Reply